Periodic table number meanings7/27/2023 
If the number were negative (Let's say 2-), then you would say "there are two less protons than electrons" and:Ģ - (-2) = 2 + 2 = 4 so there would be four electrons in this case. Take the number of protons (from the upper left) and use this:ġ+ means there is one more proton than electron. That means there is one more proton than electron. This number may be positive or negative (you could have a 3- there, instead of a 3+, for example). This may be blank - just think of a blank spot meaning "0." If it is not blank, the number there is how many more protons than electrons that the atom has. The upper right corner has to do with charge and number of electrons. and Mendeleevs justification for a periodic table based on the elements. We see that our atom has 2 protons, so we just subtract: 4 - 2 = 2 neutrons. An element is a substance whose atoms all have the same number of protons. Use the top left to find the number of neutrons. Image result for electron configuration periodic table. This number corresponds to the number of protons plus the number of neutrons. As we learned earlier, each neutral atom has a number of electrons equal to its number of protons. actinoid element, also called actinide element, any of a series of 15 consecutive chemical elements in the periodic table from actinium to lawrencium (atomic numbers 89103). Now look at the upper left corner, where the "4" is. This is the atomic number, or the number of protons. Let's start with the lower left corner, where the "2" is. The square has four corners, and sometimes there will be numbers in those corners, like:Įach of these spots may have a number that tells you something: Now imagine a little square around the He. Imagine the atomic symbol of an element, say Helium, He. If the atom is neutral (not an ion), then there are the same number of electrons as protons. That number is the "atomic number" and is equal to the number of protons in the atom. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.If you're asking this question, I'm sure that you have access to a periodic table of the elements somewhere (if not, there's one at that you can look at) If you look on the periodic table, you will see that there is a number by each element. 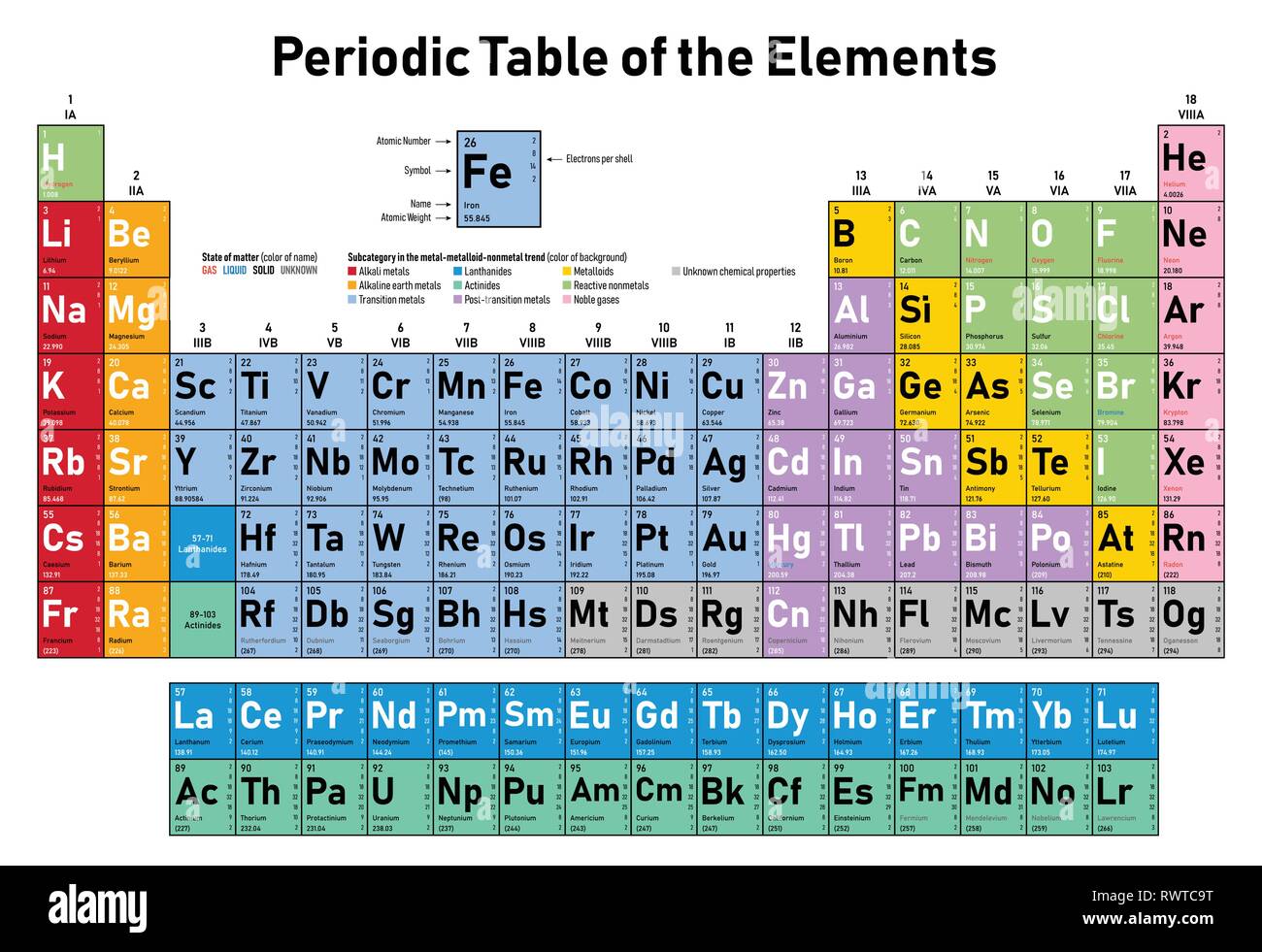
Please note that the elements do not show their natural relation towards each other as in the Periodic system. The first chemical element is Hydrogen and the last is Ununoctium. Atomic Number: the number of protons in the nucleus (which is the same as the number of electrons in the atom). Here's a close-up look at the carbon square from the Periodic Table. This means they react with other elements in similar ways. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. Elements in the same column are in the same group. This list contains the 118 elements of chemistry. Separation and Concentration Purification RequestĬhemical elements listed by atomic number The elements of the periodic table sorted by atomic numberĬlick on any elements name for further chemical properties, environmental data or health effects.Electron configurations of the next two elements in the periodic table. Plant Inspection & Process Optimalisation Thus, it takes three quantum numbers to define an orbital but four quantum.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
